Enhancing electrocatalysts by spin state modulation and external magnetic field
Green Molecules Conversion
Magnetic Field-Enhanced Oxygen Evolution Reaction via Spin Polarization Tunning
The spin state of the metal in electrocatalysts influences the catalytical properties of the catalysts, in terms of its efficiency and selectivity. Previous studies have concluded that the spin state affects both the thermodynamic interaction between the catalyst and the reaction intermediate and the kinetics of electron transfer. Based on this foundation, catalysts can be designed to lead the electrocatalytic reactions into a more efficient and selective pathway. With the assistance of external magnetic field, this optimization can be further amplified.
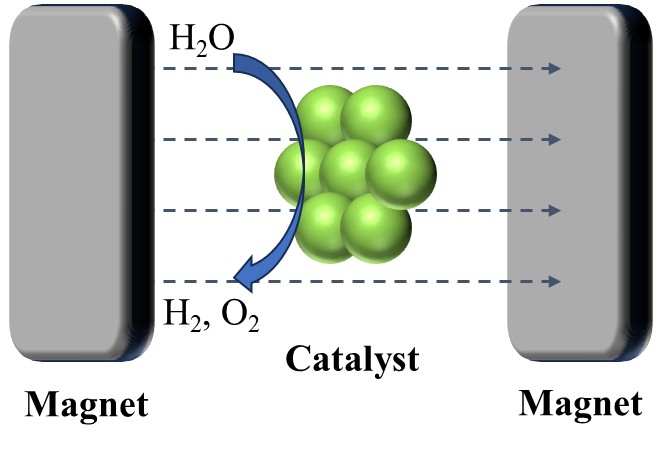
Water splitting produces H2, as a means of energy storage for clean energy. The efficiency of this process is still not ideal compared to traditional batteries. However, hydrogen energy is still an important route to the aim of net-zero emissions. As part of the water splitting process, oxygen evolution reaction is usually the rate determined reaction, due to its four-electron transfer mechanism and high kinetic barriers. Interestingly, O2 is a paramagnetic gas, making it possible to steer the reaction pathway by catalyst-metals with different spin states. By studying the mechanism behind it, more cost-efficient catalysts can be achieved for water splitting.



