Design of 3D-Printable Polymers Based on Regional Resources
Metal-free Latent Catalysts For Vitrimeric Polyesters
Foto: AI generated (OpenAI)
The Innovations Team B7 is dedicated to incorporating the vitrimeric nature into materials by developing metal-free latent catalyst systems that are activated at high temperatures. This strategy aims to facilitate efficient recycling processes without compromising the polymers’ inherent properties. Our primary focus is directed on polyesters and polyester amides, widely used in various applications, such as structural materials, fibers, or cable insulation. By fine-tuning the activation energy during the reprocessing-step via control of the catalytical activity, we aim to create an environment where these materials can be reprocessed and recycled without considerable deterioration of their properties, in turn enabling multiple life-cycles. We are further exploring reprocessing methods through 3D printing technology, enabling the creation of a more complex thermoset design via extrusion-based printing, opening new avenues for advanced material applications.
Latent Vitrimeric Reshaping of Polyesters: Capped Amines and N-Heterocyclic Carbenes as Triggered Catalysts
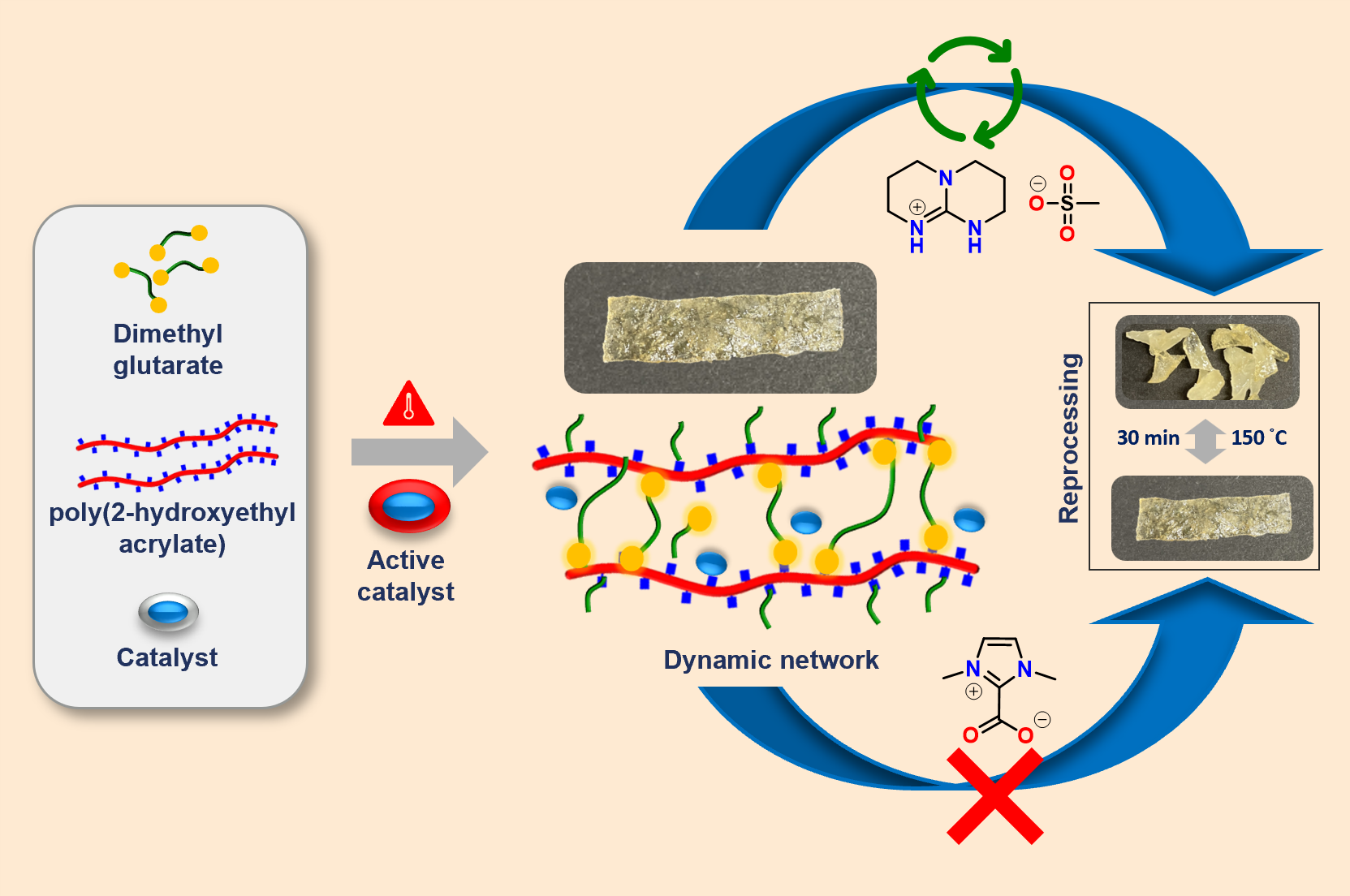
We report latent catalysts for a thermally triggered transesterification in polyester vitrimers, enabling a triggered dynamic bond exchange so offering a versatile platform for reprocessable polyesters. The blocked catalysts, such as N-heterocyclic carbenes (NHC–CO2) and the organic amine salt, triazabicyclo[4.4.0]dec-5-ene (TBD)-methanesulfonic acid (MSA), TBD:MSA, are inactive under service conditions but active at elevated temperature or shear to enable a triggered, reversible covalent bond exchange. Catalyst activation was validated in model reactions with the salt TBD:MSA as an excellent candidate to promote transesterification at 150 °C with activation energies of 34–36 kJ·mol–1 as probed by melt-rheology inside a vitrimer composed of poly(2-hydroxyethyl)-acrylate (PHEA, Mn = 68.7 kDa) and dimethyl glutarate (DMG). Mechanical testing confirmed that both pristine (E = 0.03 MPa) and reprocessed (E = 0.11 MPa) samples retained tensile properties, demonstrating efficient recyclability. In contrast, the blocked N-heterocyclic carbene (NHC–CO2) allowed an only one-time triggered activation, indicative of permanently crosslinked networks after its initial catalytic activity.